A New Covid vaccine at last
Great news, A New Covid vaccine from Valneva produces a stronger immune response when compared to AstraZeneca.
The French company reports, with no severe cases of Covid-19 seen in either group.
And new positive research on lateral flow tests. Plus guest Graham Easton discusses the urgent need for teaching climate and environmental health in medical schools.
The Health Check team consists of Presenter: Claudia Hammond Producer: Erika Wright
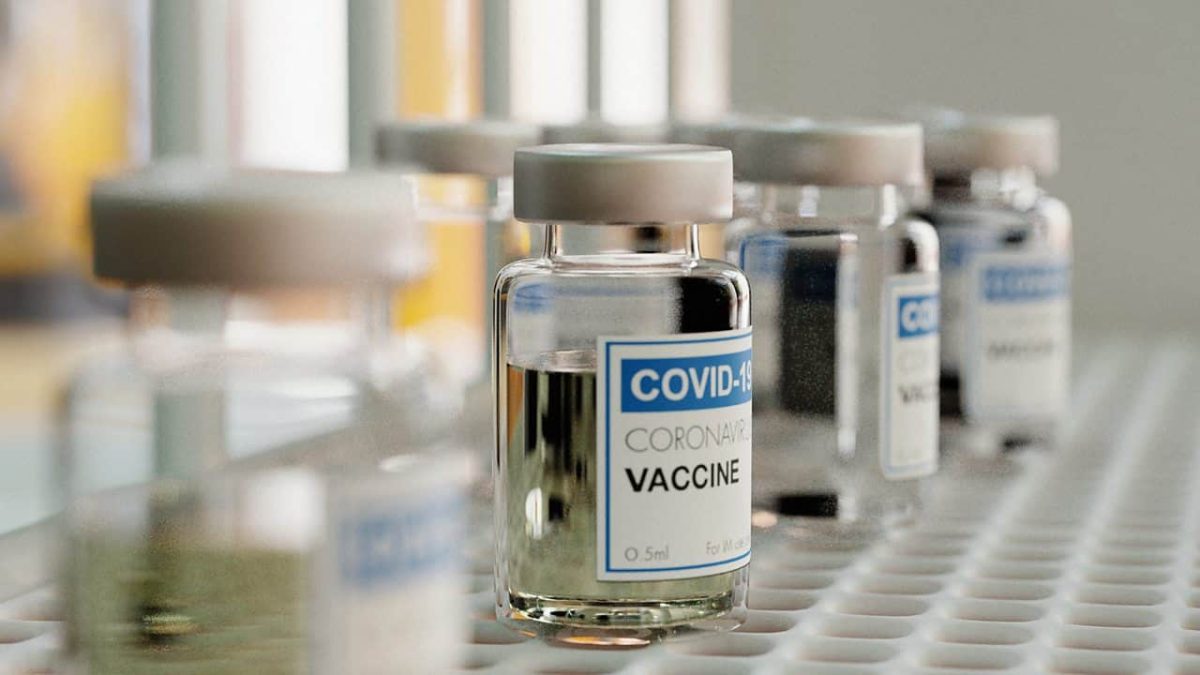
(Picture: Coronavirus vaccine vials on a laboratory shelf. Photo credit: Joao Paulo Burini/Getty Images.)
Further information on this report.
Valneva is a speciality vaccine company focused on the development and commercialization of prophylactic vaccines for infectious diseases with significant unmet medical needs. Our focused pipeline includes the only Lyme disease vaccine candidate in clinical development today, a single-shot chikungunya vaccine candidate, and an inactivated vaccine candidate against COVID-19.
The FT stated earlier this week:
The first data from the phase 3 trial showed that participants who were given the Valneva vaccine had more neutralising antibodies than those who received the AstraZeneca shot.
Valneva’s Covid-19 vaccine elicits a stronger immune response with far fewer side effects than the Oxford/AstraZeneca jab, according to trial results published a month after the UK terminated a €1.4bn deal with the French vaccine maker.
FT.Com
Meanwhile, Reuters reported in PARIS, Oct 18 (Reuters) – Valneva SE (VLS.PA) on Monday said its experimental COVID-19 vaccine demonstrated efficacy “at least as good, if not better” than AstraZeneca’s (AZN.L) shot in a late-stage trial comparing the two, with significantly fewer adverse side effects.
A question this week on Health Check, Should we mix and match Covid vaccines. New evidence from Sweden and France on the benefits of mixing and matching doses of different types of Covid vaccine.
More from the BBC
Other shows from the BBC Sunday Mornings on-air and online at 91.5 FM and 102.5 FM and available at our On-demand all week listening service.